HIV and Liver Disease
RHSP previously found that the prevalence of significant liver fibrosis was higher among HIV- infected individuals (17%) than uninfected persons (11%).
Illustraion of a normal Human Liver
About the Study
The HIV-accelerated liver disease study assessed the etiology of liver disease and HIV associated
liver fibrosis using liver biopsy.
The study enrolled participants with liver stiffness measurements >9.3 kPa
based on transient elastography scores or elevated liver function tests consistent with liver disease
Study Participants
One hundred and forty three (143) HIV- positive and 45 HIV-negative participants were enrolled of whom 113 HIV-positive and 31 HIV-negative had liver biopsy.
Biopsies were histologically staged and graded but there was no correlation between elevated liver stiffness and biopsy proven liver pathology. No active Hepatitis C virus (HCV) was detected in Rakai ( Mullis et al. CID, 2013 ) and HBV was 5% in HIV+ persons (Redd et al AIDS Res and Hum Retrovir,2013)
HBV Incidence Findings
In a retrospective cohort analysis, we investigated the incidence and risk factors associated with HBV among HIV-infected adults and found HBV incidence was significantly lower with ART use (0.49 /100 py) compared with (2.3/100 p-y) without ART [aHR=0.25 (95% CI, 0.1-0.5) p<0.001].
HBV incidence also decreased with HIV RNA suppression: (0.6/100 p-y) with ≤400 copies/mL and 4.0/100 py)with >400 copies/mL [aHR= 6.4(2.2-Clinical Studies 19.0), p<0.001] and with age: 15-29 years vs 40-50 years [aHR=3.2 (1.29.0)]; 30-39 years vs 40-50 years [aHR=2.1(0.9-5.3)] ( Seremba et al, AIDS 2017 )
Rakai Neurology Cohort Study (RNCS)
About the RNCS
Under a NIH grant we have established the Rakai Neurology Cohort Study (RNCS), which enrolled 400 HIV+ ART naïve individuals and 400 HIV-negative controls to evaluate neurocognitive comorbidity associated with HIV infection and to determine whether it varies by HIV subtype.
Baseline Statistics
- Prevalence of HIV Associated Neurocognitive Disorder (HAND) was 59% in the HIV+ individuals.
- HIV Associated Dementia (HAD) was 15% in HIV+ and 4% in the HIV- participants.
- Peripheral neuropathy was 19% in the HIV+ and 7% in the HIV- individuals (Saylor et al. in press).
The HIV infected participants were initiated on ART and followed up over 2 years.
Findings
HAND stage improved for 32% of persons on ART and dementia declined to 5%, comparable to the Clinical Studies prevalence in uninfected individuals.
However, 17% of HIV+ persons on ART had a deterioration in their HAND stage (Sacktor et al. CROI Seattle, 2017).
Immunologic studies of cerebrospinal fluid (CSF) in ART naïve HIV+ persons showed an increase in inflammatory cytokines and neurodegenerative markers in patients with lower CD4 counts and dementia. IL-6 was identified as the main driver of CNS inflammation (Abassi et al. J Neurovirol, 2016).
Phylogenetic analyses suggest a higher prevalence of HAND with subtype D (60%) than subtype A (43%.) Preliminary data suggest that 50% of HIV+ ART naïve patients have CSF compartmentalization consistent with virologic replications in the CNS.
HAND stage improved for 32% of persons on ART and dementia declined to 5%, comparable to the Clinical Studies prevalence in uninfected individuals.
Cardiovascular Disease
Illustraion of a normal Human Liver
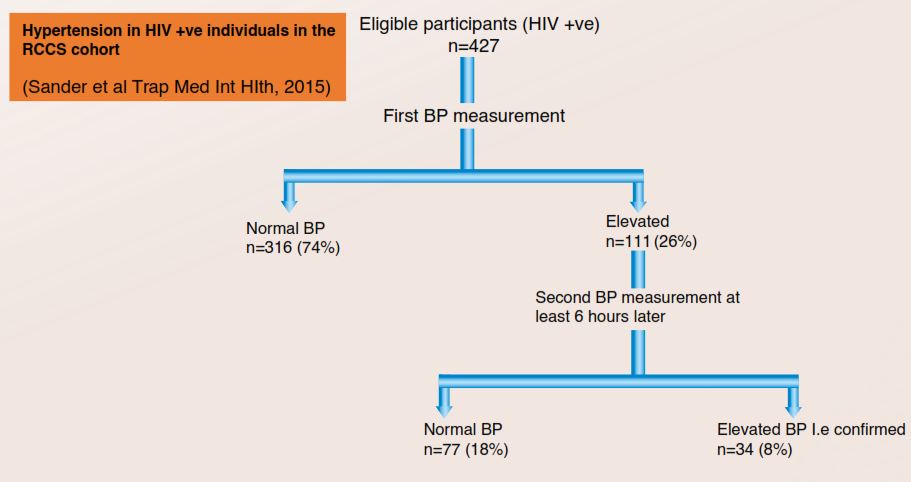
The prevalence of hypertension (two elevated blood pressures at different times) was 8% and the prevalence of elevated blood pressure (elevated at only one time) was 26%.
Participants
427 HIV+ individuals were assessed for hypertension.
Related Article
Results of Early Virologic Monitoring
Background
Viral load (VL) monitoring is standard of care in HIV-infected persons on ART. We evaluated the predictive value of VL measurements at 6 and 12 months after initiation of firstline ART to estimate the future risk of virologic failure (VF).
Methods
HIV-infected persons with VL measurements at 6 and 12 months post-ART initiation and at least 2 additional VL measurements thereafter were assessed for risk of future VF, defined per World Health Organization guidelines.
VL at 6 or 12 months post-ART was categorized into <400, 400–1000,1001–2000, and >2000 copies/mL. Cox proportional hazard models were used to compare VF incidence associated with 6-month, 12-month,and a composite of 6- and 12-month VL (Figure 1).
Results
Overall, 1863 HIV-infected adults had a 6- and 12-month VL measurement, and 1588 had at least 2 additional VLs thereafter. At 12 months post-ART, 90% had VL <400 copies/mL, 3% had 400–1000 copies/mL, 2% had 1001–2000 copies/ mL, and 5% had >2000 copies/mL
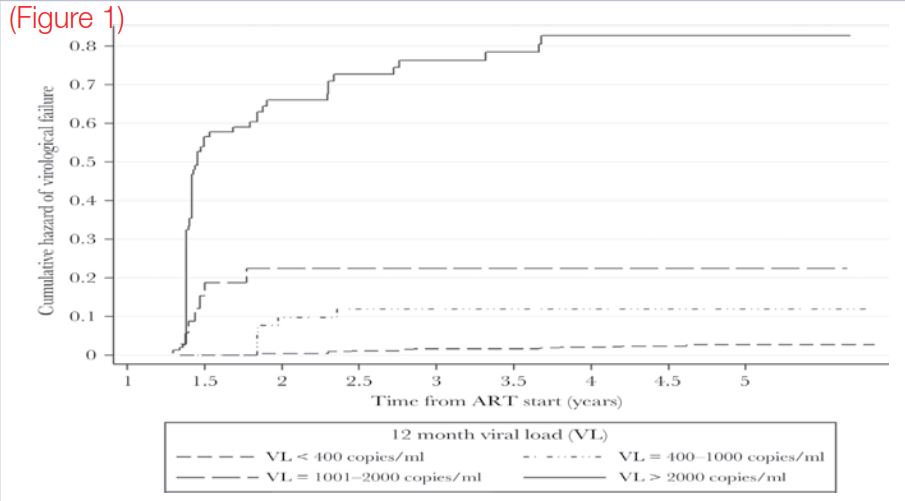
The predictive value of the 12-month VL measurement was comparable to the composite of both the 6- and 12-month (Figure 1)VL measurements and better than the 6- month VL measurement.
Conclusions
At 12 months after ART initiation, 90% of patients were virally suppressed with a low likelihood of future VF. VL measurement at 12 months post– ART initiation predicts risk of VF and could inform differentiated virologic monitoring strategies.



